We Know JAK About Myelofibrosis
Myelofibrosis treatment hasn't changed much for a long time. Is there a breakthrough on the horizon?
Of all the types of cancers, blood cancers have perhaps seen the most meaningful therapeutic advances. People often joke that blood cancers are the “the good cancers”, since a large chunk of them are quite curable or manageable long-term. Chronic Myeloid Leukemia (CML) patients have normal life expectancy because of the advent of tyrosine kinase inhibitors like GLEEVEC. Chronic Lymphocytic Leukemia (CLL) is well managed with BTK inhibitors. Acute Lymphoblastic Leukemia (ALL) and pretty much all types of B-cell lymphomas are curable with chemo-immunotherapy or CD19-based CAR-T therapy. Multiple Myeloma has steadily seen increases in overall survival with discovery of novel targets, new modalities (CAR-T and T-cell engagers) and multi-drug regimens, so much so that many leading key opinion leaders feel myeloma is entering its chronic disease era.
It has truly been one of the most optimistic areas of cancer research. I think that is why everyone really loves going to the annual American Society of Hematology (ASH) meeting every year. You’ll regularly see new data from novel MOAs that have response rates in the 90’s and complete response rates in the 70s and 80s.
That certainly isn’t the case for every hematologic malignancy. Drug-knowers will realize I omitted some pretty meaningful diseases from that list, notably acute myeloid leukemia (AML) and myelofibrosis (MF), which the rest of this post is about. Not every cancer your blood can get has yet to benefit from the major advancements our field has been able to materialize in advanced therapeutics.
But it has to be coming soon right? It must just be market forces that haven’t trickled their way down to the smaller blood cancers yet. Lymphomas and myelomas are sizable competitive markets, but those same step-change innovations we brought forth should certainly find their footing in MF, right? After all, Lilly (of all companies) just bought a company developing an MF drug for up to $2.3B
The Prolonged JAKAFI Era
Early on in my BioPharma career, I spent a great amount of my time focused on blood cancers, of which myelofibrosis (MF) was one of them. Back when I was covering this space, there was only one game in town - JAKAFI. JAKAFI is Incyte and Novartis’ JAK inhibitor which truly revolutionized treatment for MF. Before its arrival in 2011, treatment was largely reactive and palliative, relying on non-specific agents like hydroxyurea that often failed to address the core symptoms of the disease. It provided the first consistent way to significantly reduce massive splenomegaly (enlarged spleen) and debilitating constitutional symptoms like night sweats, bone pain, and extreme fatigue, which drastically improved patients' quality of life. But more importantly, it was the first to show an overall survival benefit.
🩸 What is myelofibrosis? 🩸
For the uninitiated: myelofibrosis is a rare blood cancer where the bone marrow ( aka the factory that produces your blood cells) gradually gets replaced by scar tissue. It starts with a mutation, most commonly in the JAK2, CALR, or MPL genes, that causes a hematopoietic stem cell to go rogue and proliferate uncontrollably. These malignant clones, particularly the megakaryocytes (cells that produce platelets) they produce, release a flood of pro-inflammatory and pro-fibrotic signals (think TGF-beta and PDGF) that essentially instruct the bone marrow's structural cells to lay down scar tissue where healthy marrow used to be. As the fibrosis spreads, the marrow loses its ability to make enough healthy red blood cells, white blood cells, and platelets. The spleen steps in to try to compensate, becoming massively enlarged in the process. The result is a disease defined by debilitating fatigue, severe anemia, a painful distended abdomen, and a life expectancy that, for high-risk patients, can be as short as one to two years. Allogeneic stem cell transplant is the only potentially curative option, but most MF patients are older and simply cannot tolerate the procedure Even among those who do undergo transplant, treatment-related mortality, relapse risk, and the burden of long-term immunosuppression to manage graft-versus-host disease remain significant.
As is the case with so many markets in BioPharma, a new idea doesn’t often get pursued in a vacuum. Other players (Gilead included) looked to advance slightly more refined version of JAK1/2 inhibitors to improve on JAKAFI’s profile.
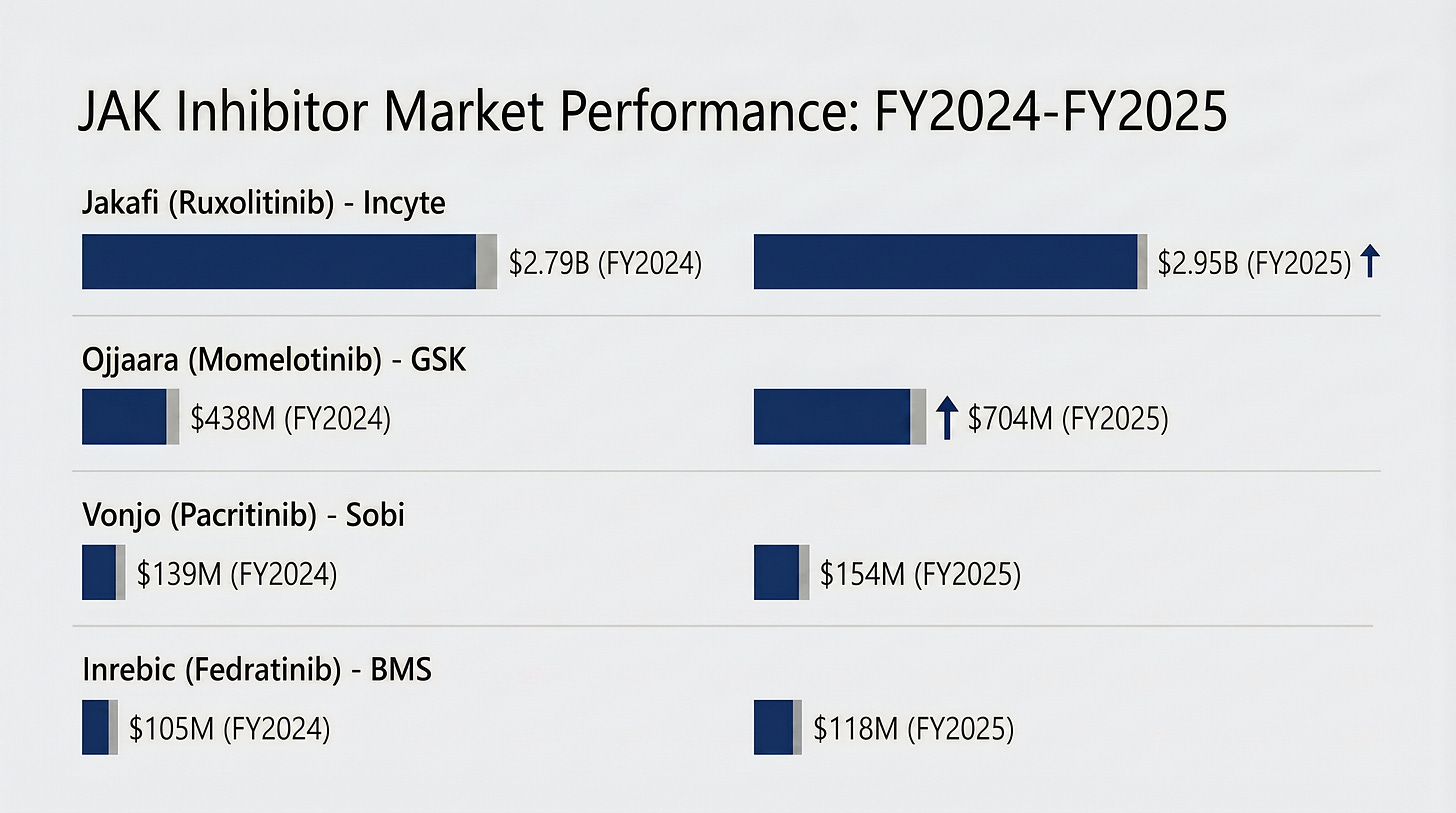
However, it took almost 8 years for new JAK inhibitor to make it to market. While JAKAFI displayed great symptom resolution (TSS) and spleen reduction (SVR), it tended to cause very high rates of anemia, which would often require patients to get blood transfusions. Treatment with next-gen JAK inhibitors from competitors shifted towards moderating SVR to spare red blood cells and reduce the blood transfusion requirements. Now there are four JAK inhibitors approved (all the ones you see in the table above) all with their particular niche in the market place.
Despite all this competition, JAKAFI still reigns supreme, but is facing a growing threat from OJJAARA, which has quickly become the go-to treatment for patients with anemia. This is a classic case of first-mover advantage paying off. JAKAFI has had significantly more time on market than its competitors, more physician experience with its product, the most robust survival data, and the benefit of still being the preferred medicine in treatment guidelines, and also the preferred treatment on formularies. But trouble may be on the horizon. Generic ruxolitinib entry is now estimated as early as December 2028, and Incyte has already faced patent litigation from generic challengers. The extended-release formulation approval is one defensive move, but whether it drives meaningful patient switching before generics arrive is an open question.
All that being said, when I put together the MF landscape for this post, I was surprised to see that the entire approved treatment portfolio is still just varieties of JAK inhibitors.
The Late-Stage MF Graveyard
However, it’s not like other strategies weren’t tried. MF’s history has some recent failures in its memory:
PI3K-delta inhibitors: This class was studied in combination with JAKAFI to enhance response. JAK inhibitors are great at reducing symptoms and shrinking the spleen, but do not slow or reverse marrow fibrosis or offer a path to cure. Incyte had its own program in this vein (parsaclisib), but it’s pivotal study was stopped early due to the added benefit over JAKAFI alone being too small to justify the added risk of thrombocytopenia.
Luspatercept (activin receptor ligand trap): Also known as REBLOZYL, this drug was aimed at reducing transfusion dependence. Unlike in MDS where it has proved to be successful, luspatercept failed to a show an improvement in this metric in MF patients.
Apoptosis pathways: Along the same vein as PI3K-delta, companies looked to target the p53 apoptotic pathway via MDM2 and BCL-2 inhibitors to add to JAK1/2 inhibition efficacy. AbbVie pushed all the wya into two Phase 3 studies with navitoclax (BCL-2 inh) but ultimately scrapped development in MF after one of its studies was unable to show a benefit in TSS, despite a significant benefit in SVR. Kartos and Novartis each advanced their own respective MDM2 inhibitors. Novartis ended up deprioritizing it’s asset, while Kartos’ navtemadlin continues on in a P3 study in 1L MF after mixed results in a P3 study in 2L+ MF.
Other approaches: A couple more that have had disappointing results: Hedgehog inhibitors, most notably Pfizer’s glasdegib, was terminated due to lack of efficacy and GI toxicity issues. Novartis tried an anti-TIM3 antibody, pursuing an immuno-oncology approach to myelofibrosis, but that was also terminated due to lack of efficacy.
Common theme across these tried-and-failed approaches is toxicity issues in combination with JAKAFI, commonly myelosuppression. The bar for success is unusually high in myelofibrosis as well. JAKAFI was approved showing improvements in total symptom score and spleen volume reduction. Many of the failures in this space have been able to show spleen volume reduction, but have failed to show a significant improvement in symptoms. While the combinatory drugs are helping to kill malignant cells more effectively, they come with additional adverse events canceling out the symptomatic relief patients feel.
More mechanistically, prolonged use of JAKAFI and other JAK inhibitors can downstream lead to mutations that render the baseline JAK backbone ineffective. The JAK problem is hard to avoid, just given how central the JAK-STAT pathway is to the pathology of myelofibrosis. However, thinking has shifted on what the appropriate endpoints for novel therapies in this space should be. The field is moving away from the “spleen-centric” era where SVR and TSS were essentially surrogate measures of efficacy; now the next wave of novel therapies are focused on showing hard outcomes like overall survival, demonstrating bone marrow fibrosis reduction (BMF grading), and reduced burden of mutant clones (Variant Allele Frequency, VAF). This offers some newfound hope to a new set of therapeutic strategies being pursued in early/mid-stage clinical development.




